Translating scientific breakthroughs into first-in-class treatments
Novel insights into human biology define our development programs that target cancer
Pipeline Overview
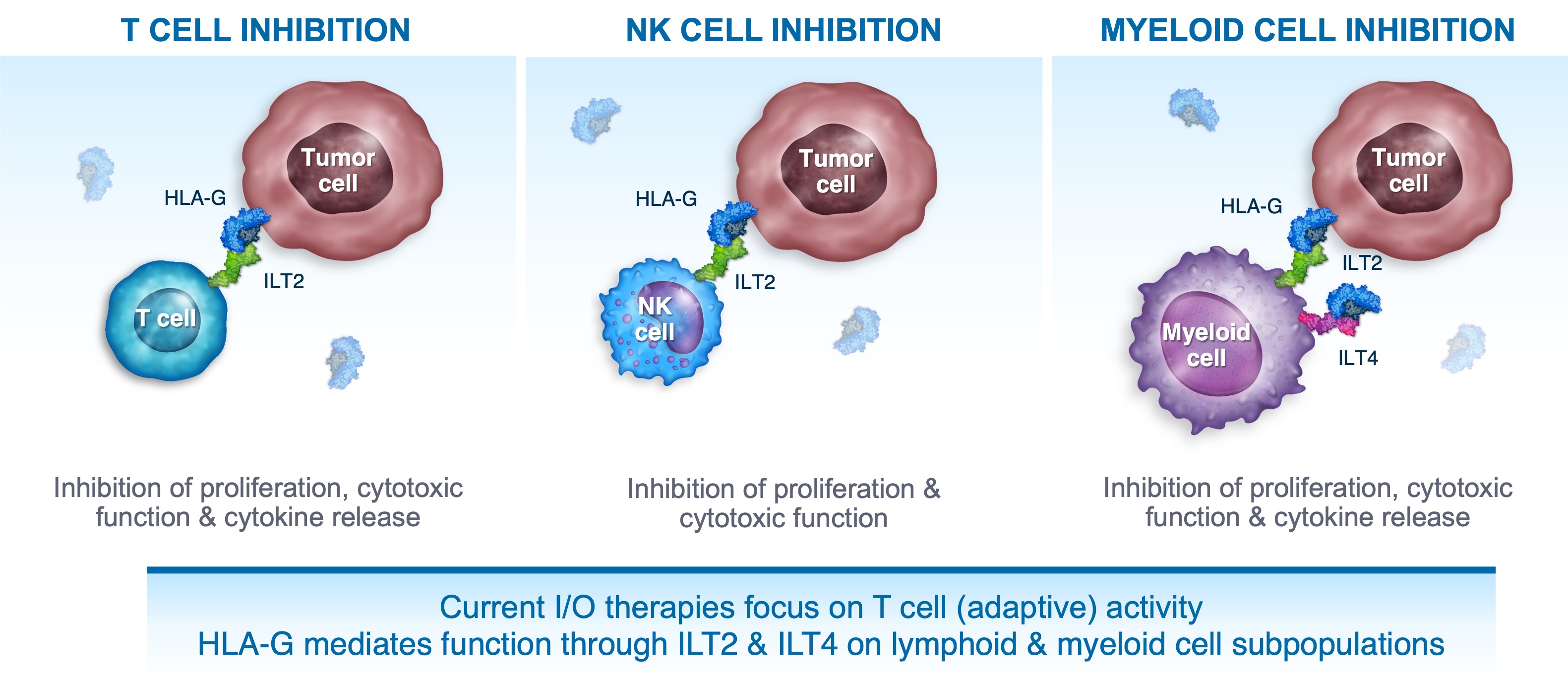
Our oncology pipeline is focused on restoring and bolstering immune responses. Tizona is enrolling patients in two randomized arms within the ongoing Phase 1b clinical trial (NCT04485013) to evaluate either TTX-080 plus cetuximab and FOLFIRI or cetuximab and FOLFIRI alone in biomarker-defined metastatic colorectal cancer.


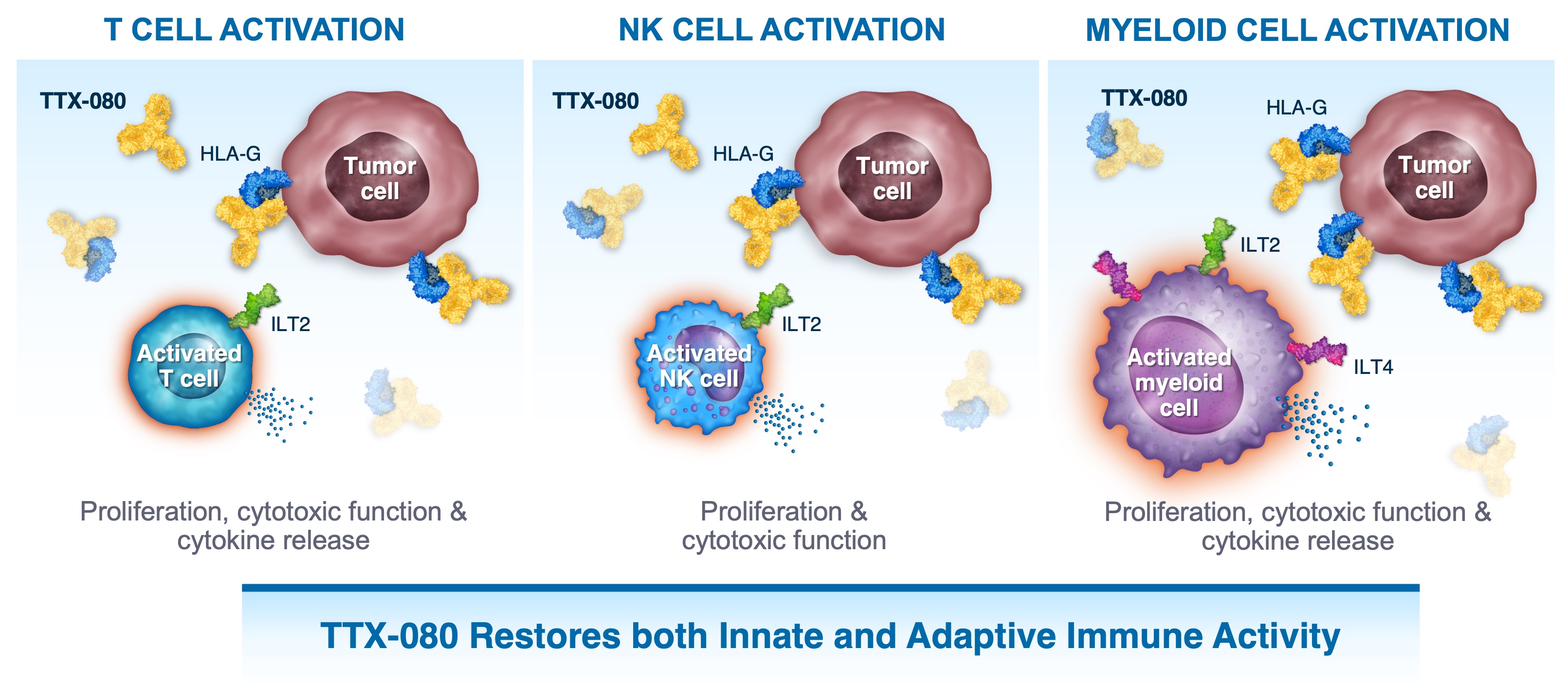
TTX-080 has the Potential to Enhance Anti-Tumor Responses
Publications
Journal for ImmunoTherapy of Cancer – February 15, 2026
Unlocking the promise of innate biology through the HLA-G/ILT2/ILT4 pathway
Society for Immunotherapy of Cancer Annual Meeting 2025
TTX-080, A First-in-Class HLA-G Specific Antagonist, Increases Distinct Innate and Adaptive Immune Cells in the Tumor Microenvironment and Periphery
American Society of Clinical Oncology 2024
Results from Ph1a/1b Analyses of TTX-080, a First-in-Class HLA-G Antagonist, in Combination with Cetuximab in Patients with Metastatic Colorectal Cancer and Head and Neck Squamous Cell Carcinoma